The research in the NESSA-Lab revolves around the study of supramolecular and colloidal self-assembly under kinetic control and dissipative - chemically fuel driven - conditions. Recent work is focussed on the fuel-controlled self-assembly of porous supramolecular materials studied under diffusion-controlled conditions.
Kinetic Control
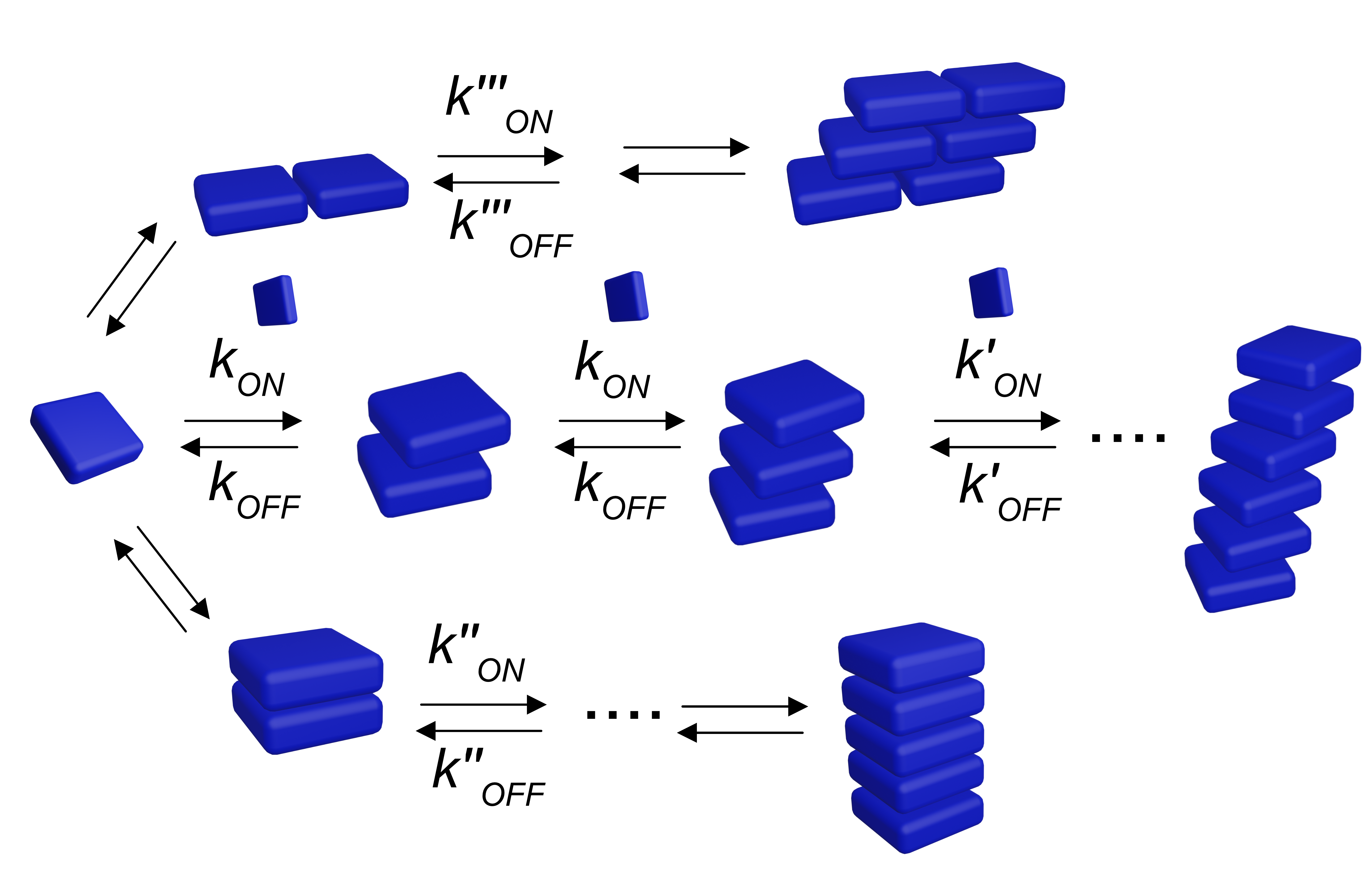
Molecular self-assembly often involves multiple competitive growth pathways featuring non-linear kinetics and different kinetic mechanisms. In addition, it occurs in a far from homogeneous and constantly evolving environment, where concentration gradients, local chemical inhomogeneities, as well as fluid dynamic and static properties impact the final outcome of the assembly process. Our aim is to understand this pathway complexity, and to control the interaction between the assembling system and its surroundings by means of different methodologies (e.g., regulating the supply and mixing of reactants and controlling reaction-diffusion phenomena in space and time). The final goal is to obtain supramolecular materials with different structures and functions but formed from the same building units, thus enlarging the toolbox of noncovalent synthesis.
Dissipative self-assembly
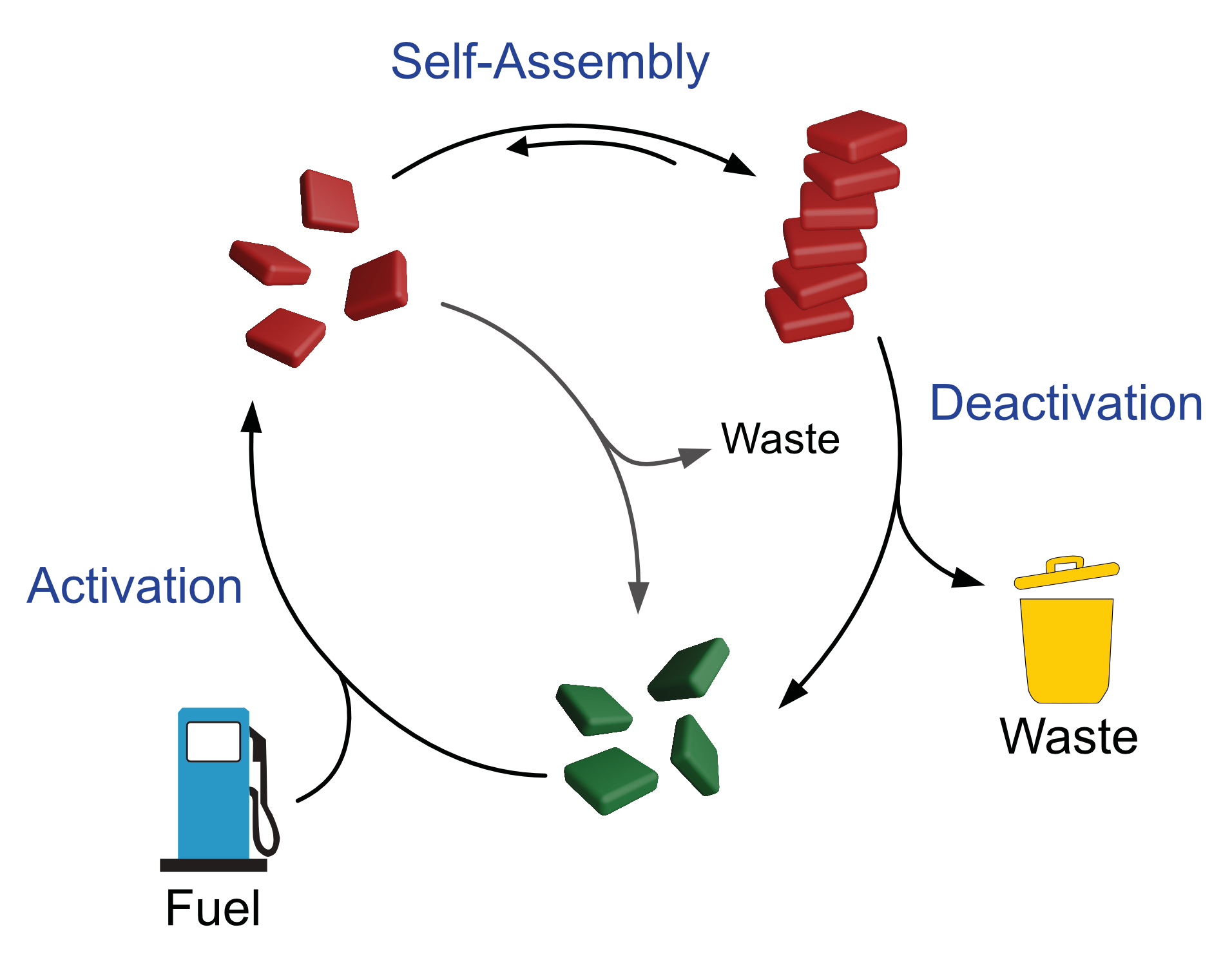
Dissipative self-assembly is the hallmark of living systems, which continuously consume energy in the form of chemical fuels to keep their functional supramolecular structures away from thermodynamic equilibrium. Under dissipative conditions, the composition of the self-assembled system is not dictated by the relative thermodynamic stabilities of the assembled and disassembled states, but rather by the dynamics of the coupled reaction network. This opens the way to emergent properties that are not achievable under equilibrium conditions. Our goal is to apply the concept of dissipative self-assembly to novel functional self-assembled materials, going beyond proof-of-concept supramolecular systems. In particular, we focus on multicomponent self-assembly, and on using compartmentalised open conditions which enable continuous supply of chemicals fuels and removal of waste.
